Example - 06 OLI Projects
Navigation: User Guide ➔ Example Projects ➔ 06 OLI
Related Links: OLI Overview
OLI Example projects have been updated in Build 139.37030 : The OLI databases used in these example projects are created with OLI Chemistry Wizard 12.0. These projects require OLI Engine Runtime (32-bit) 12.0.0.xx or newer to open.
Simple Reactor Example
Project Location
..\SysCADXXX\Examples\06 OLI\OLIFull\OLI Simple Reactor.spf
The project has been updated in Build 139.37043.
Features Demonstrated
- The use of OLI Model Configuration
- The use of OLI Reactor 2
- The use of OLI Feeder
- The use of OLI SideCalc
Brief Project Description
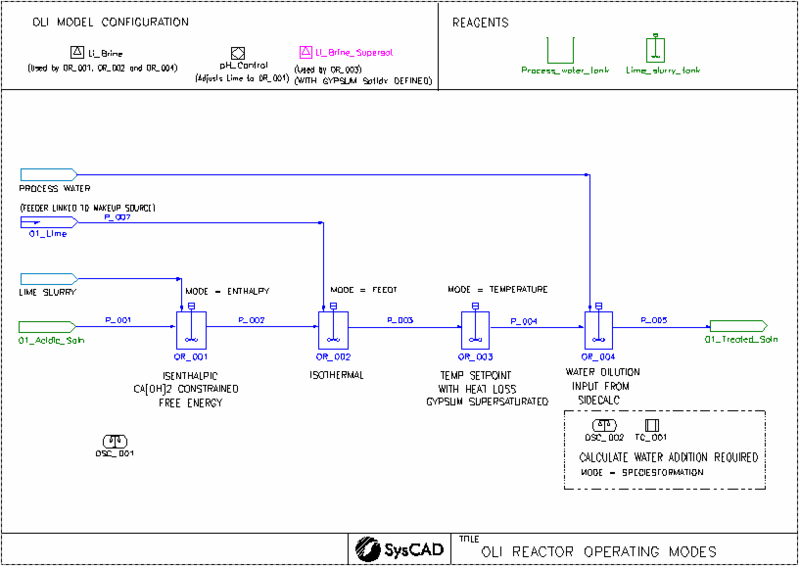
Each OLI project must contain at least one OLI Model Configuration model. We have used two OLI Model Configuration models in this project, one we allow supersaturation of gypsum.
When building a TCE project, one common uncertainty is whether the feed composition is at equilibrium conditions. Here are a few ways to check this:
- Add a side calculator (OLI Side Calc) to evaluate the stream once, then use the results as input into an OLI Feeder.
- Add an OLI Reactor and set this unit to use FeedT OpMode, with the CalcEnthalpyBal option set to OFF.
- Utilise an OLI Feeder and input the feed using OLI speciation. The OLI Feeder will then reverse map the OLI species into SysCAD species.
When using the OLI reactor model, there are different operating modes you can select. Some modes would require a single OLI equilibrium calculation while others would require iteration to achieve the results. This example project will show what these different modes are and when they should be selected.
- OpMode = FeedT: The equilibrium calculation will be isothermal, with the output temperature equal to the feed temperature.
- OpMode = Enthalpy: The equilibrium calculation will be isenthalpic, with the output temperature calculated. Use this mode where species composition changes and the delta energy change is important. For example, use this for all the reaction tanks where heat is released due to acid-base reactions.
- OpMode = Temperature: The equilibrium calculation will be isothermal, with the output temperature equal to the temperature setpoint.
- OpMode = Species Formation: This is demonstrated using the side calculation where a target calculation determines the water required to dissolve the gypsum entering the tank. The calculated water flow is then applied to the makeup of the actual reactor tank, demonstrating a feed-forward control strategy for water addition.
Project Configuration
- The Li_Brine database is used by most of the units.
- A second Li_Brine database (Li_Brine_Supersat) is used by the TR_012 unit to emulate the Gypsum being supersaturated.
- CaSO4:2H2O(s)(Gypsum) formation has been suppressed in the "OLIConfig" tab.
- Salts First algorithm has been selected for species re-building in both model configuration files. See Reverse Mapping section for more details.
OLI Unit models
- TR_001, TF_001 and TSC_001
- The OR_001 unit is set up to evaluate the feed using OLI speciation at the specified temperature. This is not a real reactor, so the delta heat change is not important, and the CalcEnthalpyBal option is switched off. If the stream composition is far from equilibrium and not solvable at the feed temperature, having CalcEnthalpyBal off is crucial to prevent OLI from trying to solve an unsolvable stream.
- TF_001 can be used to determine a set of SysCAD species using OLI speciation at the required temperature.
- TSC_001 checks the speciation for the lime slurry stream. If the stream composition is acceptable, no action is required.
- TR_002, TR_003, TR_004
- These are all using the enthalpy operation mode. This mode calculates the heat for each input stream, as well as the heat for the product. Therefore, for each iteration, a tank with two input streams will require a minimum of three OLI calculations. The CalcEnthalpyBal option is always on for this mode.
- Heat loss has been added to OR_004.
- TR_011, TR_011A and TR_011B
- The primary purpose of this set of reactors is to demonstrate how to configure the system to simulate limestone efficiency. Since some of the limestone may be unreactive, we can account for this by using the WithBypass option (as shown in TR_011A), which excludes the unreactive portion from the solution. When this option is enabled, the input stream used for the OLI calculation is reduced accordingly. The bypassed amount is then added back to the OLI output to form the final discharge stream. TR_011B provides a visual representation of how the WithBypass function operates.
- TR_012
- This reactor uses a different OLI chemistry model (Li_Brine_Supersat), where the formation of CaSO4:2H2O(s) (gypsum) has been suppressed in this unit.
- Heat loss has been added to this unit.
- TR_013 and TSC_002
- TSC_002 is configured to calculate the water needed to produce 12 tonnes per hour of CaSO4(s) in TR_013. This involves an iterative process where the OLI equilibrium calculation is repeatedly performed until a solution is achieved.
- The calculated water flow is then used to adjust the makeup of the actual reactor tank, showcasing a feed-forward control strategy for water addition.
Note: For information on the number of TCE calculations performed per SysCAD iteration, please refer to the Usedby tab in the TMC unit model.
Acid Recovery - Nanofiltration Example
Project Location
..\SysCADXXX\Examples\06 OLI\OLIFull\AcidRecovery_NF.spf
Available from Build 139.31866.
Features Demonstrated
- The use of OLI Model Configuration
- The use of OLI Reverse Osmosis Unit
Brief Project Description
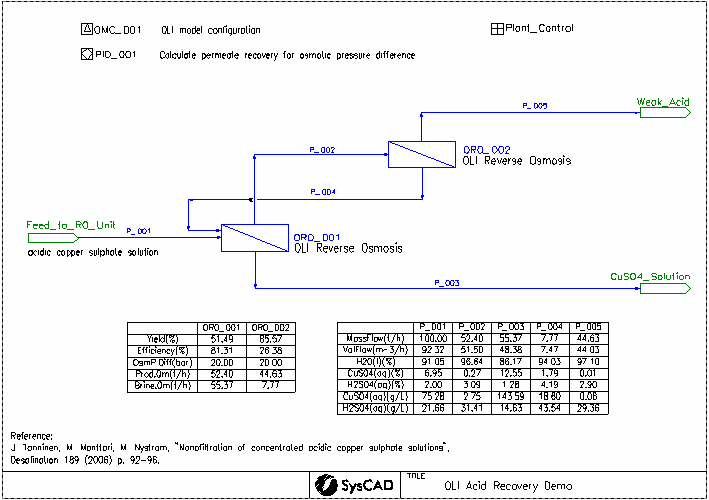
This project was created based on the paper: "Nanofiltration of concentrated acidic copper sulphate solutions". See reference below. It looks at the separation of sulfuric acid from copper sulfate at high salt and acid concentrations. OLI Reverse Osmosis Unit is used to simulate the nanofiltration units.
Reference:
- J. Tanninen, M. Manttari, M. Nystrom, "Nanofiltration of concentrated acidic copper sulphate solutions", Desalination 189 (2006) p. 92-96.
Project Configuration
- The CuSO4_H2SO4.dbs database is used. This was generated using the OLI MSE framework.
- Salts First algorithm has been selected for species re-building. See Reverse Mapping section for more details.
- OR0_001
- Efficiency Method set to Ionic Permeability.
- Yield required controlled by OsmP difference value, set to 20 bar in the PID controller.
- A concentration polarization factor of 120% is applied.
- Ion permeability is set by the Plant_Control General Controller
- OR0_002
- Efficiency Method set to Ionic Permeability.
- Yield required controlled by OsmP difference value, set to 20 bar in the PID controller.
- A concentration polarization factor of 120% is applied.
- Ion permeability is set by the Plant_Control General Controller
For both of the OLI reverse osmosis units, the following streams are calculated using OLI:
- Input: The combined input streams at equilibrium
- Output: The brine solution leaving the unit at equilibrium
- MembraneInterface: The brine solution at the membrane interface, which is more concentrated than the Output stream due to concentration polarization
- Product: The acid solution which has passed through the membrane
The osmotic pressure difference is calculated between the MembraneInterface and the Product.
Potash Crystallisation Example Project
Project Location
This is a Steady State project and is stored at:
..\SysCADXXX\Examples\06 OLI\OLIFull\Potash_Crystallisation.spf
Available from Build 139.31866.
Features Demonstrated
- The Use of OLI Model Configuration - with an OLI potash database file (potash.dbs).
- Setting up flash trains using OLI Evaporator with Shell and Tube Heat Exchanger 2 & Barometric Condenser.
NOTE: This is similar to the Potash example project, but instead of using the Potash add-on, it is using the OLI Solver.
Brief Project Description
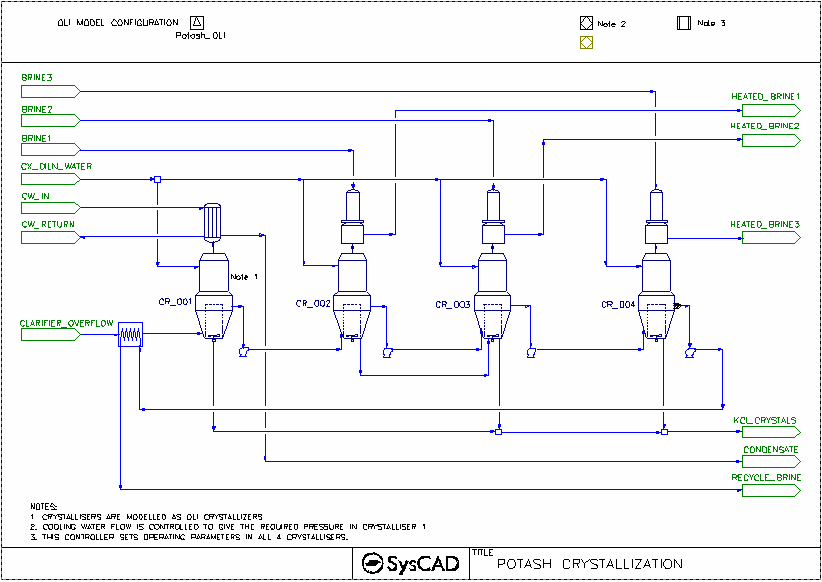
- This is a MOP (Muriate of Potash) example project.
- This project shows a 4-Stage-Evaporator(crystalliser) circuit.
- This project uses constant feed for Liquor Feed and Brine streams.
- Crystallisation is based on OLI solver solution.
- Dilution water is added to each crystallisation stage.
- PotashEvaporator underflow (crystallised material) is removed from the first third and fourth stage. Second stage underflow is feed to the 3rd stage.
- Brine is used to exchange heat in the Barometric condensers.
Project Configuration
- The potash.dbs database is used. This is generated using the OLI MSE framework.
OLI Evaporator
- OLIEvaporator Tab
- Select FlashTrain mode
- Specify minimum flash temperature for the unit (this will assist with pressure balance of the circuit)
- Set up the TCE tab page - use "Potash_OLI"
- Set up the Separ tab page - required Overflow and Underflow solids.
- Set up dilution water to the crystalliser.
Barometric Condenser
- Set the approach temperature.
Shell and Tube Heat Exchange
- HX_001 is used to condense the flash steam from the 1st Crystalliser,
- Method - CondensingSteam
- Condensing Method: Demand (UA)
- Demand Connection: FlashTrain
- The condenser area and HTC information is supplied.
- HX_002 is used to preheat the feed with recycle Brine
- Method - Sensible HX
- The heater area and HTC information is supplied.
Autoclave for Downhole Condition Testing
Project Location
..\SysCADXXX\Examples\06 OLI\OLIFull\Autoclave for Downhole Testing.spf
Available from Build 139.37077.
Features Demonstrated
- The use of OLI Model Configuration
- The use of OLI Reactor 2
- The use of simple PGM files.
- The use of a PID Controller.
Brief Description
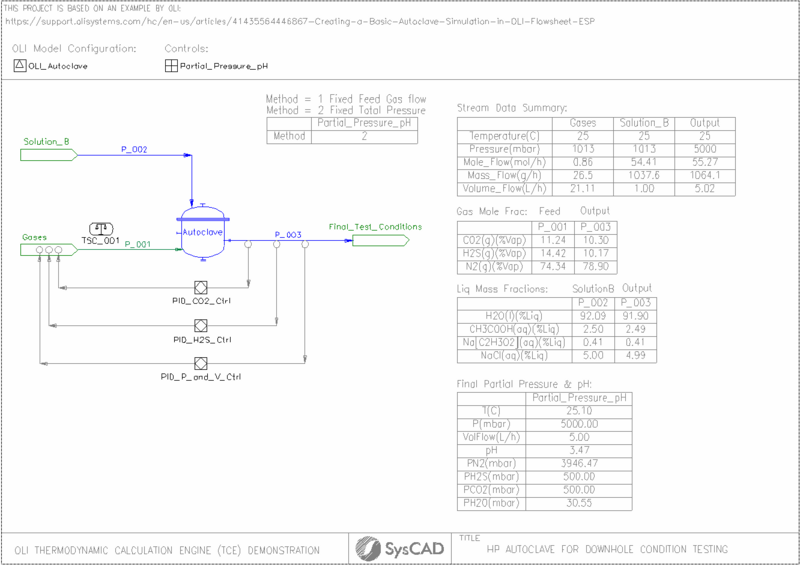
Autoclaves enable corrosion testing under high‑pressure conditions encountered in oil and gas operations. Accurately reproducing these environments is challenging, and a key requirement is preparing a gas mixture that achieves the correct partial pressures of acidic gases such as CO₂ and H₂S at the target final pressure. This project was developed in reference to an example provided by OLI, cited below, and expands on that work. The objective is to demonstrate how SysCAD, when used together with OLI, can be applied to accurately define the required gas‑phase conditions for creating the final downhole conditions. This combined approach provides a reliable method for establishing proper autoclave test setups for corrosion testing.
Reference:
- “Creating a basic autoclave simulation in OLI flowsheet: ESP,” OLI Systems, Inc, https://support.olisystems.com/hc/en-us/articles/41435564446867-Creating-a-Basic-Autoclave-Simulation-in-OLI-Flowsheet-ESP
Project Configuration
- The OLI_Autoclave.dbs is used. This was generated using the OLI MSE-SRK framework.
- Salts First algorithm has been selected for species re-building. See Reverse Mapping section for more details.
- Uses required pressure and enthalpy operating mode.
- Partial pressure of the outlet stream is calculated using results from the OLI Reactor and displayed with the pH in the Partial_Pressure_pH Controller (General Control -PGM files).
- Final partial pressure of CO2 and H2S were measured and adjusted with PID_001.
Demo OLI Lithium Carbonate Acid Leach Project
NOTE: The following example is for OLI released with 9.3 Build 137. In Build 139.31866, a new set of OLI models have been released, and the Build 137 models have been archived as Legacy. The legacy models and the following project will be discontinued in the next major release (Build 140), please change and existing projects to use the new set of models as soon as possible.
Project Location
..\SysCADXXX\Examples\06 OLI\OLILegacy\Demo OLI Lithium Carbonate Acid Leach Project.spf
Features Demonstrated
- The use of OLI Chemistry Model
- The use of OLI Reactor to perform side calculations
- The use of Thickener model
- The use of Filter Press model
- The use of Composition Fetch in Feeder model
- The use of Makeup Sources and Makeup Blocks
- The use of Reaction Blocks, including the use of Heat Exchange, Override Product Temperature Option and Final Flow extent type
- The use of Set Tag Controllers
- The use of a PID Controller
- The use of EHX
- The use of Split Flows Gas Vent option
Brief Project Description
- This is a simplified model of a Lithium Carbonate plant. The project is based on the Quebec Lithium Project in Canada, as described in "Technical Report NI 43-101 on the Pre-Feasibility Study for the Quebec Lithium Project", by Canada Lithium Corporation and BBA Inc, May 2010, which was downloaded from the Sedar website (http://www.sedar.com/DisplayCompanyDocuments.do?lang=EN&issuerNo=00007891).
- It is very similar to the Demo Lithium Carbonate Acid Leach Project except in a couple of places it uses OLI to predict reaction extents and pH.
- Four parts of the process are mimicked in OLI Reactors using the Composition Fetch functionality in the feeders and Set Tag Controller to transfer results to corresponding reaction blocks. The four processes are:
- Water Leach
- Hydroxide Precipitation
- Manganese Precipitation
- Lithium Carbonate Precipitation
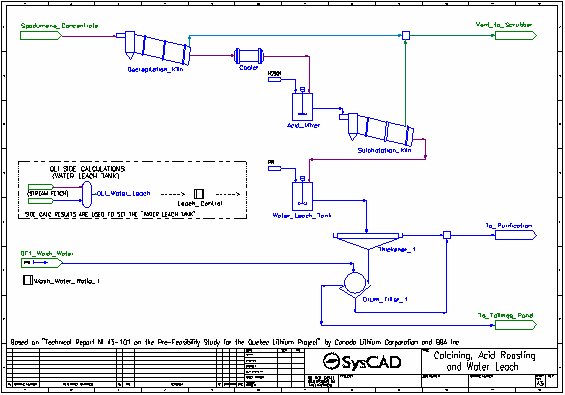
- Dried spodumene concentrate is fed into the Decrepitation Kiln, where alpha-spodumene is converted to beta-spodumene and any water present is evaporated. These reactions occur at 1050 °C.
- The solid product is cooled to 60 °C in the Cooler.
- Cooled solid product is mixed with concentrated sulfuric acid in the Acid Mixer.
- The slurry from the Acid Mixer is fed to the Sulphatation Kiln where the beta-spodumene is converted to solid lithium sulfate by reacting with the sulfuric acid. Oxide impurities are also converted to their sulfate forms by reaction with sulfuric acid. These reactions occur at 250 °C.
- Some excess water is converted to gas. All gases are vented.
- The kiln solids, along with leftover acid, pass to the Water Leach Tank where water is added.
- The majority of the reactions in the Water Leach Tank are set by the Leach Control controller which measures the results generated by OLI in the OLI Water Leach OLI Reactor.
- The slurry is thickened and send to a drum filter where it is washed with process water. The washed filter cake is sent to the Tailings Pond.
- The filtrate and thickener overflow are combined and sent to the purification processes.
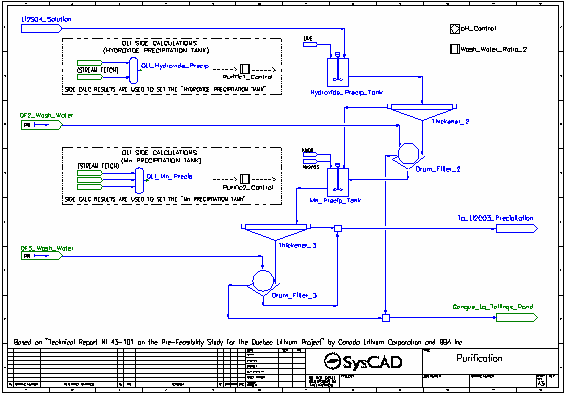
- The pH of the lithium sulfate solution is raised from around 3 to around 6 by the addition of Lime. The pH is calculated by the corresponding OLI Reactor, OLI Hydroxide Precip, and the addition of the Lime is controlled by a PID Controller.
- The Lime neutralises most of the acid present as well as participating in precipitation reactions.
- The majority of the reactions in the Hydroxide Precip Tank are set by the Purific1 Control controller which measures the results generated by OLI in the OLI Hydroxide Precip OLI Reactor.
- The resulting slurry is thickened and send to a drum filter where it is washed with process water. The washed filter cake is sent to the Tailings Pond.
- The filtrate and thickener overflow are combined and sent to the second stage of purification.
- The pH of the solution is raised to 10 by the addition of Caustic Soda (NaOH). The pH is calculated by the corresponding OLI Reactor, OLI Mn Precip, and the addition of the NaOH is controlled by a PID Controller.
- The NaOH both neutralises all remaining acid present as well as participating in precipitation reactions. Soda Ash is also added to aid in the precipitation of carbonates.
- The majority of the reactions in the Mn Precip Tank are set by the Purific2 Control controller which measures the results generated by OLI in the OLI Mn Precip OLI Reactor.
- The resulting slurry is thickened and send to a drum filter where it is washed with process water. The washed filter cake is sent to the Tailings Pond.
- The filtrate and thickener overflow are combined and sent to the lithium carbonate precipitation tank which is maintained at 95 °C.
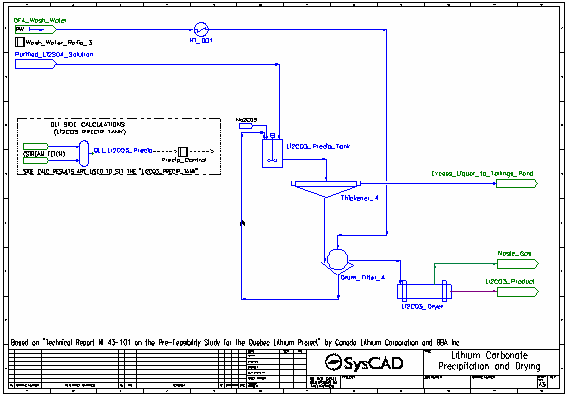
- Soda Ash is added to convert the lithium sulfate to lithium carbonate (which precipitates), as well as precipitate other carbonates.
- The majority of the reactions in the Li2CO3 Precip Tank are set by the Precip Control controller which measures the results generated by OLI in the OLI Li2CO3 Precip OLI Reactor.
- The resulting slurry is thickened and send to a drum filter. The thickener overflow is sent to the Tailings Pond.
- The thickened slurry is washed with process water (also at 95 °C) in the drum filter. The washed filter cake is sent to the dryer, while the filtrate is recycled back to the lithium carbonate precipitation tank.
- In the dryer, all water is evaporated and aqueous species are returned to their solid forms. The steam is vented leaving the solid lithium carbonate product.
Project Configuration
- Most units with reactions are simulated by the Tank model. This includes the Decrepitation Kiln, Sulphatation Kiln, Water Leach Tank, Hydroxide Precip Tank, Mn Precip Tank, Li2CO3 Precip Tank and Li2CO3 Dryer.
- Four OLI Reactors are used to perform side calculations (using the Side Calc model). The results used include pH and final solid flows to set dissolution and precipitation reactions.
- The reactions set based on OLI predictions use the Final Flow extent type.
- Separation of gases and slurry is usually achieved by the use of Split Flows Gas Vent option.
- Makeup Blocks are used:
- in the Acid Mixer to add a slight excess sulfuric acid
- in the Water Leach Tank to add process water to achieve a specified solids fraction
- in the Hydroxide Precip Tank to add a controlled mass flow of lime solution
- in the Mn Precip Tank to add a controlled mass flow of NaOH solution
- in the Mn Precip Tank to add a stoichiometric amount of Na2CO3
- in the Li2CO3 Precip Tank to add a stoichiometric amount of Na2CO3
- The Cooler is modelled by a Tank with the EHX sub-model enabled to achieve the desired product temperature
- All Thickeners are modelled by the Thickener model using the OverFlowSolidsFraction method.
- All Drum Filters are modelled by the Filter Press model using the SolidsFractionInFiltrate and Constant Wash Efficiency methods.
- Set Tag Controllers are used to set wash water flows based on user specified ratios to all drum filters and to set final flows of species in reaction blocks (based on OLI predictions).
- PID Controllers are used to control the Lime and Caustic Soda additions in order to achieve the desired pH, based on OLI calculations of pH.
Included Excel Report
Lithium Acid Leach Example Report.xlsx
This file has the following reports:
- Criteria
- Streams
- Mass Balance