Reaction Block - HOR & Energy
Navigation: Models ➔ Sub-Models ➔ Reaction Block (RB) ➔ Heat of Reaction (HOR), Energy, Dilution, Partial Pressure
| Overview | Reaction Block Data Section | RB Sub Model (Model Theory) | ||||||
|---|---|---|---|---|---|---|---|---|
| Reaction Editor | Reaction Block Summary | Reaction Block Tabs (Main) | Individual Reactions | Reaction Extents | Source / Sink / Heat Exchange | Solving Order - Sequential or Simultaneous | Energy Balance | Heat of Reaction / Heat of Dilution / Partial Pressures |
Heat of Reaction
By default the Heat of Reaction (HOR) is calculated from the difference in the sum of the heats of formation of the products and reactants. (Please see Reaction Block Energy Balance for a description of an energy balance around a reaction block.)
The enthalpy of the product stream will be calculated as follows:
- Enthalpy of Product Stream at Product Conditions = (Enthalpy of Feed Stream at Feed Conditions) - (Heat of Reaction at Reference Conditions)
The Heat of Reaction at Reference Conditions (0°C and 101.325kPa) is calculated either using the heats of formation of the reactants and the products in the Species database, or the user may specify a Heat of Reaction. These two methods are described in the sections below.
Note:
- The user may display the Heat of Reaction values at 0°C, Feed Temperature, Product Temperature and at a User Defined Temperature. Please see Display Additional Reaction Information .
Use Heats of Formation
This is the default method used by SysCAD to calculate the HOR.
Heat of Reaction at Reference Conditions = (Total Enthalpy at Reference Conditions for products) - (Total Enthalpy at Reference Conditions for reactants)
- where Total Enthalpy = Heat of Formation at 25 + [math]\displaystyle{ \int\limits_{25}^{T}C_p dT\, }[/math]
- The Heats of Formation at 25°C are defined in the Species database.
- The Cp values, or equation, is also defined in the Species Database.
User Specified Heat of Reaction
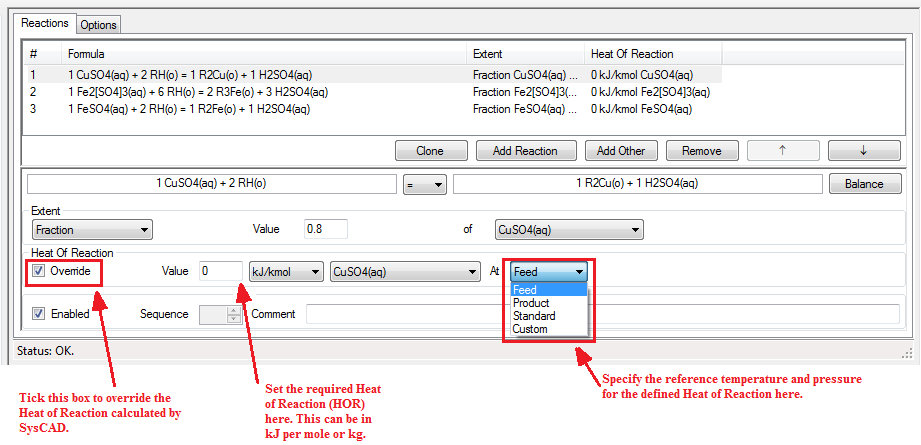
The user can override the default HOR calculation and specify a Heat of Reaction in the reaction file by enabling the Override tickbox for Heat of Reaction for the specific reaction. The following options are then available:
- The Heat of Reaction may be specified as either kJ/kg or kJ/mol.
- The user specifies the species (reactant or product) to use for the heat of reaction calculations.
- The user specifies the reference Temperature and Pressure for the user defined Heat of Reaction. The options available are:
- Feed - the Feed temperature and pressure is used;
- Product - the Product temperature and pressure is used;
- Standard - Standard conditions (25°C and 101.325kPa) are used; or
- Custom - The user may specify the temperature, in °C, and pressure, in kPa, for the specified HOR.
Notes:
- If the user wishes to override the heat of reaction such that the temperature will not change (i.e. Tfeed = Tproduct), then the heat of reaction should be set to 0 at the Feed conditions.
- If the user wishes to override the heat of reaction such that the sensible heat (HS) will not change (i.e. HS_feed = HS_product), then Custom should be used with the HOR value = 0 at 0°C and the unit pressure.
- Similarly, if the user wishes to override the heat of reaction such that the sensible heat (HS) will change by a fixed amount ΔHS (i.e. HS_feed = HS_product + ΔHS), then Custom should be used with the HOR value = ΔHS at 0°C and the unit pressure.
- Ideally the user should try obtain HOR reference data at 0°C and use the Custom option to specify the HOR at 0°C.
- If the user defined Heat of Reaction is specified at any temperature other than 0°C, then SysCAD will adjust it to 0°C in either of the following ways:
- If the Standard species model is used then SysCAD will use the Cp data in the species database to calculate HOR at reference conditions.
- If a Bayer Properties or any user defined species model is used, then SysCAD will use the enthalpies of the feed and product streams from the reaction to calculate the HOR at reference condtions.
Example of a User Defined Heat of Reaction:
Heat of Dilution in Reaction Energy Calculations
SysCAD uses Heat of Formation (Hf) and Specific Heat (Cp) data to calculate Heats of Reaction (HOR). However, for species with Heat of Dilution data, e.g. H2SO4(aq), Hf is a function of the mass fraction of the species in the solvent, in most cases H2O(l). There are a number of methods of dealing with this in the Reaction Block, but a single methodology, as described below, has been chosen:
- H2SO4(aq) is used as the example species in this example.
- The feed to the reaction block contains H2SO4(aq) with fraction = Feed Fraction (which could be zero).
- For all reaction HOR calculations in the reaction block, the Hf of H2SO4(aq) = Enthalpy at infinite dilution, i.e. fraction of H2SO4(aq) = 0.
- After all reactions have been solved, the final product stream from the reaction block contains H2SO4(aq) at fraction = Product Fraction (which could be zero).
- The Heat of Dilution is then calculated after all reactions have taken place (this is shown as [email protected](aq) on the RB access window) This calculation has two parts which must be added together:
- The HOR for H2SO4(aq) at Feed Fraction → H2SO4(aq) at infinite dilution. If there is no H2SO4(aq) in the feed then this term is zero.
- The HOR for H2SO4(aq) at infinite dilution → H2SO4(aq) at Product Fraction. Again, if there is no H2SO4(aq) in the product then this term is zero.
- If the Heat of Dilution override is selected then [email protected](aq) = 0.
Note: Also refer to Heat of Dilution.
Partial Pressure of Gases in Reaction Energy Calculations
- The enthalpy of some gaseous species can vary with pressure (e.g. H2O(g) (steam)). Its enthalpy is calculated using its partial pressure which is a function of the total pressure and its molar fraction relative to other gases.
- There are many ways that this could be dealt with in the Reaction Block, but a single methodology has been chosen as described below (H2O(g) is used as the gaseous species in the following example):
- The feed to the reaction block contains H2O(g) with PP = PPFeed (which could be zero).
- The user specifies a method to use for calculating the partial pressure to be used (PPUsed). By default, this will be PPFeed.
- For all reactions in a reaction block, the Hf to be used in the HOR calculations for H2O(g) will be its enthalpy at PPUsed. (i.e. PPUsed is used for calculating Hf of both H2O(g) reactants and H2O(g) products in all reactions)
- After all reactions have taken place, the final product stream from the reaction block contains H2O(g) with PP = PPProd (which could be zero).
- After all reactions have taken place, the Work ([email protected]) must be calculated. This has two parts which must be added together:
- Work.Feed: The HOR for H2O(g)@PPFeed → H2O(g)@PPUsed. Obviously if there is no H2O(g) in the feed then this term is zero.
- Work.Product: The HOR for H2O(g)@PPUsed → H2O(g)@PPProd. Obviously if there is no H2O(g) in the product then this term is zero.
There are several methods available to the user for calculating/setting the partial pressure used when calculating the enthalpy of gaseous species to be used in the reaction block calculations. Use of the different methods will not change the amount of material reacted or the final temperature of the stream. It will only change where the energy is reported between the HOR for an individual reaction and the Work term. The methods available are as follows:
- No Partial Pressure Override: The partial pressure used will be the feed partial pressure.
- Total Pressure Method: The partial pressure used will be the total feed pressure.
- Feed PP Method: The partial pressure used will be the feed partial pressure.(This is identical to not using the Partial Pressure Override).
- Product PP Method: The partial pressure used will be the product partial pressure.
- Enviro PP Frac Method: The partial pressure used will be the environmental fraction of the gas (as defined on the Plant Model-Environment page) multiplied by the total pressure.
- User Defined PP Frac Method: The partial pressure used will be the user defined fraction of the gas multiplied by the total pressure.
- User Defined PP Method: The partial pressure used will be the user defined partial pressure.
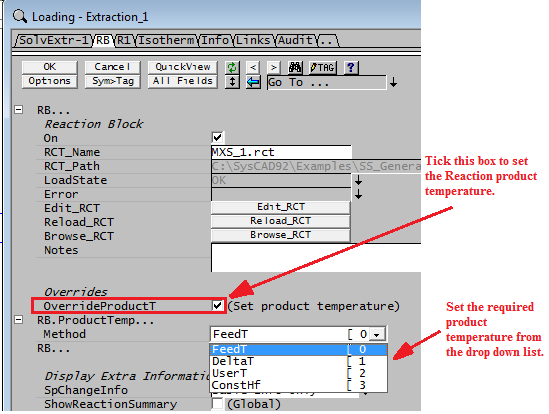
Override Product Temperature Option
This option allows the user to set the product temperature and remove the requirement for SysCAD to perform and report detailed HOR calculations for the individual reactions and overall HOR. SysCAD still maintains an energy balance and reports the overall HOR and energy added (or removed) to maintain the energy balance across the RB.
The advantage of this option is that there is significantly less work for SysCAD and hence an increase in solver speed. This is an ideal option to use when forcing the final product temperature and where reporting of the energy terms such as the HOR of individual reactions is not important. It is more efficient than using the equivalent RB HX or EHX to set the final temperature.
The options for forcing the product temperature are:
- Maintain the feed temperature
- The product temperature is a fixed amount different to the feed temperature
- The product temperature is forced to a user specified value
The overall Energy Balance of the model is maintained and reported in two places. Firstly at bottom of RB page under the heading "Energy Balance (with product temperature override)"; and secondly by reporting the energy override on the Audit page. This option is similar to using a User Specified Heat of Reaction Override and using one of the Heat Exchange options such as FinalT.
Note: When this option is used many of the energy values normally visible (such as heats of reaction) are not available.